Philosophical Tours in Britain
Active in planning voyages of discovery, Cavendish never went on one himself. He did, however, make a number of journeys by carriage within Britain to expand his knowledge. On the first journey we know anything about, he passed through Oxford to Birmingham and back by way of Towcester, making trials of Edward Nairne’s
An inveterate traveler, Blagden recorded his journeys in notes and letters, beginning with a journey he took to Scotland to study at age seventeen.3 We have his report of a visit to Wales when he was twenty-three, an impressionable if unfocused tourist. An admirer of Rousseau, the “most eloquent & feeling of men,”4 he was drawn to abbeys and vistas but he was also interested in mines, ironworks, and “philosophical curiosities.” Having a strong desire to know the larger world, he was struck by the “extreme stupidity” of people who were entirely satisfied with their “little world.” Wherever he traveled he was frustrated because people could not answer his simple questions about what lay a mile around them—places, routes, departures.5 When after serving several years as a surgeon to the British Army in North America he returned to England, he toured Devonshire where he found the coves and rocks “beautiful” and “romantic,” and where he also observed mileages, weather, slate, and clay.6 His most consequential journey was from Plymouth to London in 1781, where he made a life for himself in science.
It was Blagden who suggested the journey that he and Cavendish made in 1785. Early that year he proposed that they visit John Michell
There was nothing odd about Cavendish’s curiosity about ironworks. The English aristocracy was generally forward-looking, ready to promote and invest in industry
Cavendish and Blagden kept an account of their tours, written in part by Blagden, and in part by Cavendish.10 Their first stop in 1785 was Alderley in Gloucestershire, where they stayed with Blagden’s older brother John Blagden Hale
Ever since Pascal
Writing in 1777 Shuckburgh
For carrying up mountains, Cavendish had a portable barometer made by Ramsden
Heights of Mountains

Fig. 16.1: Garth Mountain. Near Cardiff. On the lower left, we see a furnace. Courtesy of Cardiff Central Library.

Fig. 16.2: Portable Barometer. Photograph by the authors at Chatsworth. This is probably the barometer that Cavendish carried to the top of Garth Mountain to measure its height. When folded into its mahogany case, the barometer measures 43½ inches. The instrument is suspended in gimbals. At the bottom, near the wooden cistern, there is a thermometer with a corrections scale. William Roy, with whom Cavendish collaborated on experiments with barometers, used a portable barometer almost identical to this one for taking heights of mountains. Although the Chatsworth barometer is unsigned, we know from Roy that this kind of barometer was made by Jesse Ramsden. Roy (1777, facing p. 658). The photograph is reproduced by permission of the Chatsworth Settlement Trustees.
Lewis
The operations that Cavendish and Blagden witnessed centered on iron smelting
Ironworks

Fig. 16.3: Working Iron at Merthyr Tydfil. Watercolor by J.C. Ibbetson in 1792. A mass of hot iron is being struck by a trip hammer to remove slag. Courtesy of Cyfarthfa Castle Museum.
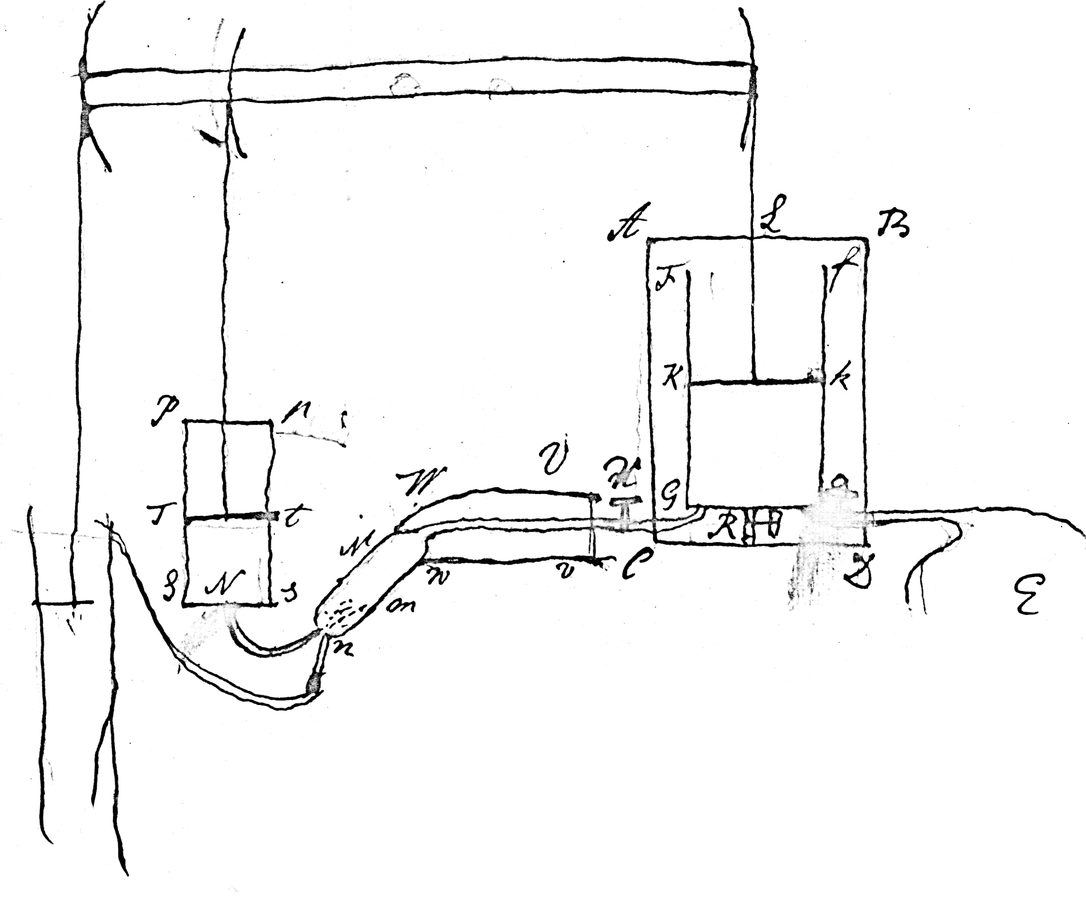
Fig. 16.4: Cavendish’s Drawing of a Steam Engine. In this diagram, Mm is the condensation chamber, Pp is the air pump, and is Ff is the working cylinder. Cavendish gives the dimensions and the strokes per minute of the engine, and he notes its advantage: “In common [Newcomen] engine as much steam condensed on sides as is used to fill the cylinder.” Cavendish Mss, Misc. Reproduced by permission of the Chatsworth Settlement Trustees.
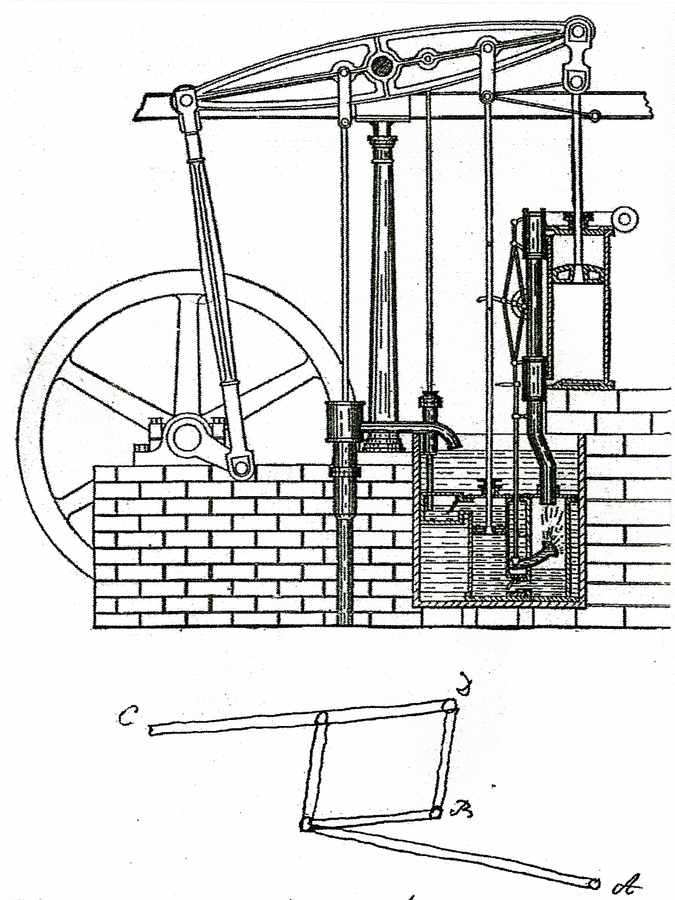
Fig. 16.5: Parallel Motion. In the early Watt engines, the piston was connected to the beam by a chain. By replacing the chain with a rod, it was possible to develop power on the upward as well as the downward stroke, to push as well as pull, doubling the action of the engine. There was a problem, however. The piston rod moved vertically, while the beam moved circularly. Watt solved the problem with a four bar linkage between the rod and the beam in the form of a familiar pantograph, which produces parallel lines; in this case, parallel motion. A piston moving vertically up and down transmitted force in both directions to a circularly moving beam. Watt took out a patent on his “parallel motion” in 1784. Cavendish drew a picture of the linkage in his 1785 journal; it is shown at the bottom of this illustration.
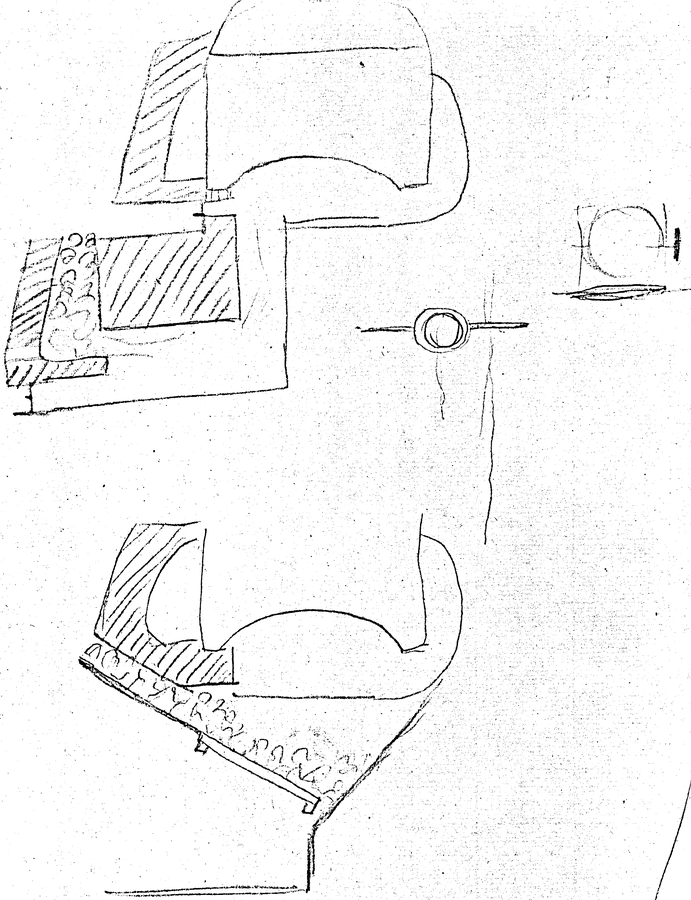
Fig. 16.6: Cavendish’s Drawing of Watts Furnace for Burning Smoke. In 1785, Watt patented a smoke-consuming furnace. It had two sources of heat. On a grate, there was a regular fire, the first source. Where the fire was drawn into a flue or chimney, there was a second grate containing red-hot coals that had ceased to smoke, the second source; there the smoke of the first fire was consumed. Cavendish Mss, Misc. Reproduced by permission of the Chatsworth Settlement Trustees.

Fig. 16.7: Albion Mills. Cavendish may have observed Watt’s smoke-consuming furnace in Birmingham on his journey in the summer of 1785, or he may have observed it at Albion Mills, located on the Surrey side of Blackfriars Bridge. Built-in 1783–86, Albion Mills was the largest and technologically most up-to-date flour mill of the time. In the fall of 1785, Watt came to Albion Mills where his steam engine was to be installed. It was his advanced double-acting, rotative engine, proper for turning mills, and it was to be worked by his newly invented smoke-consuming furnace. In 1789, a second engine was installed. In 1791, Albion Mills burned down. It bears on Cavendish’s interest that later that year, he together with Blagden, Banks, and the engineer John Smeaton were invited to inspect drawings of a steam engine and a waterwheel at Falcon Stairs, near Blackfriar’s Bridge and the former Albion Mills. Charles Blagden to Joseph Banks, 23 Oct. 1785, Banks Correspondence, Kew, 1:212. John Maitland to Joseph Banks, 19 Dec. 1791, Manuscript Department, British Museum, Add Mss 33979, p. 118. Wikimedia.
Iron production
Midway through their journey Blagden sent Banks an encouraging report. They had seen cloth and iron manufactures
New Willey Ironworks near Broseley in Shropshire was their next stop. Its ironmaster was John Wilkinson, whose innovative boring mill for making cannon was exactly what Watt needed to make accurate cylinders for his engines, improving their efficiency by correcting for leakage of steam. They visited a second, new ironworks of Wilkinson’s at Bradley, near Birmingham. This ironworks differed from others they had seen in the use of reverberatory furnaces instead of the traditional hearth forges, in which iron lies directly on the fuel, which contains impurities. The advantage of reverberatory furnaces is that the iron is separated from the fuel, heated by hot gases flowing over it and by radiant heat reflected from the roof of the furnace.
They visited the ironworks at Colebrookdale, a large plant a quarter mile in length, near the historic Ironbridge. Abraham Darby III, the third-generation head of the company, had made castings for the bridge, the first major structural use of cast iron. The still-standing 100-foot, semi-circular bridge spanning the River Severn linked ironworks at Coalbrookdale with sites across the river.20 The blast for the two furnaces at Coalbrookdale was delivered by two cylinders powered by water raised by a steam engine of Watt’s design. The year of Cavendish and Blagden’s visit another steam engine was installed to blow air at two forges located outside the building.21
Steam engines came with the setting of their journeys, which was the Industrial Revolution
Wherever they went, they talked to owners, engineers, and workman, who gave them information no one else could. In their journals they also recorded observations of strata, rocks, and pebbles surfacing the roads, and on separate sheets they kept a record of barometer and thermometer readings, from which Cavendish calculated elevations. Blagden wrote to Banks that Cavendish “bears the journey remarkably well.”22 The journey lasted about three weeks.
The following year, 1786, Cavendish and Blagden set out again on a three-week journey, this one longer than the first, to the north of England.23 They traveled directly to John Michell’s
Cavendish took advantage of the journey to follow up his chemical interests. He accepted Lord Mulgrave’s
In Sheffield they observed file-making and other manufactures
For the third straight year, in 1787 Cavendish and Blagden set off on a journey, this time to the southwestern corner of England, Cornwall. They brought with them letters of introduction written by Watt
On their route to Cornwall, they followed the seacoast “on account of particular experiments to be done there.”42 On Dartmoor in southwest Devonshire, they carried out an elaborate series of observations with barometers, thermometers, and rain gauges having to do with a problem in the barometric measurements of heights. Blagden, who had lived in nearby Plymouth, made the local arrangements, which involved the assistance of three other men and the construction of a small meteorological observatory
On their journey, between industrial sites they observed strata
Cavendish and Blagden made no more journeys together. In the summer of 1788, Blagden went to France, sending back scientific news to Cavendish.47 So familiar had they become as a traveling pair that the following year Blagden had to correct Deluc, explaining that he was planning a tour of Italy not with Cavendish but with Lord Palmerston
Such were Cavendish’s journeys in his middle years. Setting out from London in different directions, he explored different corners of the kingdom. Wherever he went, he examined industrial processes, materials, and products, determined the heights of mountains, observed the “order of the strata,” and collected stones, noting their physical characteristics and investigating them chemically. From his observations and other sources, he wrote a paper on the strata of the island.51 He was a tourist with an active curiosity and definite tastes: what interested him he pursued tirelessly, and what did not he silently ignored.
A great reader of travel books, as we know from his library, Cavendish was prepared to be enticed out of his study by Blagden and to become himself, for a time, a traveler. His journals differ from the usual types of travel journals by their exclusive focus, though they have much in common with the geological and industrial observations of William Lewis’s
The journeys marked a change in the direction of Cavendish’s work
The “White Book” came to light relatively recently. The variety of substances Cavendish examined can be suggested by a few entries: whitish sparkling ore from Hudson’s Bay, native iron from Mexico, earth from Isle of Man, lava from Mount Vesuvius, limestone, chalk, clay, and mica. Making no distinction between the natural and the manmade, the book also records experiments on specimens from mines and wastes from industrial processes such as kish from iron furnaces, slag from the purification of copper, finery cinder, and dust from lead smelting furnaces. The engineer James Cockshutt supplied Cavendish with specimens of coal and iron, and Cavendish wrote a paper on the making of iron with recommendations for the engineer,56 an exchange we might view as an early meeting of two revolutions, the scientific and the industrial.
Cavendish’s journals are the first indication of his active interest in geology. In Britain in the late eighteenth century, the main spur to geology came from what he
Weighing the World
The first indication of Cavendish’s
Michell’s apparatus came to be known as a “torsion balance
Cavendish was nearly sixty-seven when he “weighed the world,” the name he and Michell
Apparatus for Weighing the World

Fig. 16.8: Apparatus for Weighing the World. Cavendish’s modified and rebuilt version of John Michell’s apparatus. The large metal spheres R are weights that attract small metal spheres suspended from the ends of the arm, which in turn is suspended by the fine wire gl. The room in which the apparatus is housed and protected is also shown as are the arrangements for viewing it from outside the room. “Experiments to Determine the Density of the Earth,” PT 88 (1798):526.
Cavendish began his account with words that should encourage readers, “The apparatus is very simple” (Fig. 16.8). Its principal moving part was a six-foot wooden rod suspended horizontally by a slender wire attached to its center, and suspended from each end of the rod was a lead ball two inches across. The whole was enclosed in a narrow wooden case to protect it from air currents. Toward the ends of the case and on opposite sides of it, were suspended two massive lead balls, or “weights,” each weighing about 350 pounds. Cavendish rebuilt Michell’s apparatus.
The force that turns the rod aside is the gravitational attraction between the weights and the balls. From the angle of twist of the rod and the period of vibration of the rod moving freely as a horizontal pendulum, the density of the Earth is deduced. It is not obvious how the Earth enters the experiment, becoming “obvious” only when the formulas for the forces acting in the experiment are written out and the resulting equations are combined. Cavendish did not use equations but worked with proportions, and as a result his reasoning is unfamiliar to a modern reader. The experiment essentially compares the gravitational attraction of the lead weights on the balls with the gravitational attraction of the Earth on the same, the source of the Earth in the experiment.69
Earlier in the century it had been an open question whether or not a mass the size of the mountain is sufficient to cause a measurable effect. Twenty-five years before Cavendish’s experiment, the Royal Society had carried out a successful experiment on a mountain, and as we have seen, Cavendish had helped prepare for it. In the experiment Michell invented, Cavendish achieved a measurable effect with masses small enough to fit into an apparatus. Newton had been discouraging, having calculated that if two one-foot spheres of Earth-matter were placed only one-quarter inch apart, they would not “come together by the force of their mutual attraction in less than a month’s time.” Newton
Because the smallest disturbance could destroy the accuracy of the “weighing,” Cavendish placed the apparatus in a small, closed “room” about ten feet high and as many feet across. From outside the room, Cavendish worked pulleys to swing the weights close to the case to set the rod in motion, the deflection and vibration of which he observed by means of telescopes installed at each end of the room. Veniers at the end of the rod enabled him to read its position to within one hundredth of an inch. The only light admitted into the room was provided by a lamp near each telescope. Once an experiment was underway, it was not interrupted until the end; depending on the stiffness of the suspension wire, it might take as long as two and one-half hours.
Given that the apparatus was simple and the procedure straightforward, it might seem that Cavendish’s report of the experiment would be brief. It was not, taking up fifty-seven pages in the Philosophical Transactions, in length second only to his paper on the theory of electricity. The reason it was long was Cavendish’s concern with accuracy. Near the beginning of his paper, where he estimated the minuteness of the gravitational force, he began a discussion of errors and corrections, which he continued to the end. The following account gives an idea of Cavendish’s meticulous way of experimening.
Looking ahead to the conclusion, that unequal heating of the air was the disturbing force that was hardest to avoid, Cavendish explained how he located and designed the apparatus to minimize this main “source of error
Cavendish was not finished with errors. In calculating the density of the Earth from his data, he made several idealizations: the arm and the copper rods holding the weights have no weight, the weights attract only the nearest ball, and the attraction of the case is ignorable. In light of these, he made six “corrections,” five of which were not of “much signification,” but were “not entirely to be neglected” either. The important correction was the effect of the position of the arm on the attraction between the weights and the balls, which influenced the time of vibration. One of the corrections, that of the effect of the mahogany case on the arm inside it, required an extensive analysis, which Cavendish included in the paper as a mathematical appendix, even though the “whole force is so small as not to be worth regarding.” In the conclusion of the paper, Cavendish gave a table of results of the seventeen experiments. They agreed closely with one another, but still the differences were too large to be explained fully by the “error of observation” or by air currents owing to temperature differences. He expressed the final outcome of the experiments as a mean of the results for each of the two wires, finding the two means to be the same. Noting that the extreme results differed from the mean by no more than one fourteenth of the whole, he concluded that the mean density of the Earth was determined “to great exactness” as 5.48 that of the density of water.72
Cavendish thought that his readers might object that because the outcome was influenced by currents of heated air, it could be influenced by yet another source, “some other cause, the laws of which we are not well acquainted with,” leading to “a considerable error in the result.” To put to rest this objection, he reminded his readers that he had made the experiments in various weathers and temperatures. He anticipated another objection; “namely, that it is uncertain whether, in these small distances, the force of gravity follows exactly the same law as in greater distances.” His reply was that there was no evidence that the law differs “until bodies come within the actions of what is called the attraction of cohesion, and which seems to extend only to very minute distances.” Nevertheless he carried out a number of experiments with the balls placed as close to the case as possible, finding no difference. In these ways, Cavendish concluded his paper with second and third thoughts about possible factors affecting the accuracy of the outcome.
The experiment of weighing the world consisted of observations of matter moving in response to two of the best-known forces, gravity and the restoring force of twisted wire, but as we have seen, to achieve accuracy, Cavendish had to consider nearly all of the forces known to natural philosophy: in addition to gravity and elasticity, they were forces associated with magnetism, electricity, deformation, heat, and cohesion. Cavendish’s mastery of the art of experiment rested on his mastery of natural philosophy.
Despite and in part because of his last experiment Cavendish had not freed himself from the claims of the earlier method of determining the density of the Earth, the attraction of mountains. His paper brought a prompt response from Charles Hutton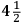 , in the “calculation of which” he, Hutton, had borne “so great a share.” Anyone who has looked at Hutton’s laborious calculations can sympathize. Hutton thought that Cavendish’s wording hinted at inaccuracies in his calculations and seemed to disparage the Royal Society’s experiment on the mountain in Scotland. That experiment, Hutton reminded Cavendish, had determined not the density of the Earth but only the ratio of that density to the density of the mountain, 9 to 5. Hutton had supposed that the density of the mountain is the density of ordinary stone,
, in the “calculation of which” he, Hutton, had borne “so great a share.” Anyone who has looked at Hutton’s laborious calculations can sympathize. Hutton thought that Cavendish’s wording hinted at inaccuracies in his calculations and seemed to disparage the Royal Society’s experiment on the mountain in Scotland. That experiment, Hutton reminded Cavendish, had determined not the density of the Earth but only the ratio of that density to the density of the mountain, 9 to 5. Hutton had supposed that the density of the mountain is the density of ordinary stone,
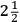 times that of water, but the actual density of the mountain was unknown, as Hutton had pointed out at the time. All that was known was that Schehallien was a “massive stone,” and Hutton now believed that its density was higher, 3 or even
times that of water, but the actual density of the mountain was unknown, as Hutton had pointed out at the time. All that was known was that Schehallien was a “massive stone,” and Hutton now believed that its density was higher, 3 or even
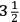 , which would make the density of the Earth “between 5 and 6,” where Cavendish had put it, and “probably nearer the latter number.” The Royal Society had not finished its experiment because it had not determined the density of stone, Hutton said. Even now, he hoped that the Society would do it, so that “an accurate conclusion, as to the density of the earth, may be thence obtained.”74
, which would make the density of the Earth “between 5 and 6,” where Cavendish had put it, and “probably nearer the latter number.” The Royal Society had not finished its experiment because it had not determined the density of stone, Hutton said. Even now, he hoped that the Society would do it, so that “an accurate conclusion, as to the density of the earth, may be thence obtained.”74
Cavendish, as we have seen, repeated his experiment many times, in different seasons, and with attention to a range of possible errors and corrections, and he had taken mean values, considered the spread of the extreme values, and in general estimated the confidence that could be placed in 5.48. At the bottom of Hutton’s letter to him, Cavendish drafted a brief response, which is identical to the last paragraph of his published paper.75 In that paragraph, Cavendish did not commit himself as to which density, his or the Royal Society’s, was more to be “depended on,” since the Society’s was “affected by irregularities whose quantity I cannot measure.”76
In 1811, the year after Cavendish’s death, John Playfair
In the next century, the astronomer Francis Baily
Weighing the world had a precedent in William Gilbert’s
Cavendish had assisted the Royal Society in preparing the experiment on the mountain, but he did not take part in the experiment. His own experiment with metal spheres, his gravitational terrellas, corresponded to his normal way of life. To weigh the world, he did not need to go out into it; he could do it, and do it more precisely, in his laboratory, using an apparatus and reasoning from universal principles. He stayed at home and looked inside of the room and through a slit in a case, inside of which was the world on his terms.
At his home on Clapham Common, he worked largely in seclusion, though he used assistance when he needed it; in the last two parts of his experiment on the density of the Earth, he had George Gilpin
Weighing the world has been called a beautiful physics experiment, but it would be incorrect to call Cavendish a physicist, as we understand the word. He was a natural philosopher of the eighteenth century. One of the differences between the two is the conditions of work. In 1878 John Henry Poynting
When physics emerged in the nineteenth century, the worldview of physical science had changed from from Cavendish’s day. An example is the role of time
The Cavendish Experiment
John Playfair
Writing to Banks in 1802, Blagden
In time the Cavendish experiment ceased to be regarded as a way to determine the density of the Earth, even as it continued to be performed. It became instead the experiment to determine “big G,” the gravitational constant appearing in the law of gravitational force, defined as the strength of attraction between two one-kilogram masses one meter apart. As C.V. Boys
The Cavendish experiment today is often called the experiment to determine G, which is correct given that the experiment is the common possession of physics. It is often said that Cavendish’s object was to determine G, which as a historical statement is incorrect but understandable given that the constant is more significant than the density of the Earth. In Cavendish’s time, there was no independent unit of force, such as our dyne and Newton. The strength of any force was expressed in terms of an equivalent gravitational attraction, and weight was the measure of mass. The universal gravitational constant did not come up, though we can easily calculate it from Cavendish’s data.89 We find implicit in his work two of the three principal universal constants
Today, 300 years after Newton and 200 years after Cavendish, gravity is still at the center of physical research. To quote from a publication by researchers in the field: the “most important advance in experiments on gravitation and other delicate measurements was the introduction of the torsion balance by Michell and its use by Cavendish …. It has been the basis of all the most significant experiments on gravitation ever since.”90
By its method and example, Cavendish’s experiment has had a far-reaching influence on physics. In “Cavendish’s skillful hands,” the torsion balance has “revolutionized the science of precision measurement”; not only have nearly all of the determinations of G been done with that instrument, but it has been used in “countless other applications, such as seismological measurements and electrical calibration—wherever precise control over very small forces is called for.”91 A contributor to a symposium on general relativity traces the “noble tradition of precise measurement to which we are heirs” to Cavendish’s experiment, which he calls the “first modern physics experiment.”
Footnotes
Henry Cavendish, “Trials of Nairne’s Needle in Different Parts of England,” Cavendish Mss IX, 11:45–54. Dates in the second half of August 1778 are scattered through this record of observations.
George Hunt to Mr. Hext, 23 Jan. 1787, Blagden Papers, Yale, box 1, folder 4.
Charles Blagden to Sarah Nelmes, 1 Nov. 1765, Blagden Letters, Royal Society, B.159. In other letters from 1767 Blagden gave Nelmes accounts of shorter journeys in Scotland. Nelmes, who lived in Bristol, was related to Blagden. “Accounts, Bills, Insurance, and Copy of Will of S. Nelmes,” Blagden Mss, Royal Society.
Blagden recommended reading Rousseau to Thomas Curtis, 26 July 1771, Blagden Letters, Royal Society, B.162.
Charles Blagden, “Memorandum of a Tour Taken for Four Days Beginning August 18 1771,” Blagden Papers, Yale, box 1, folder 3.
Charles Blagden, “Tour of the South Hams of Devonshire,” 1780, Charles Blagden Diary, Yale, Osborn Shelves f c 16.
Charles Blagden to John Michell, 25 Apr. 1785 and 13 Sep. 1785, drafts; in Russell McCormmach (2012, 399).
Charles Blagden to William Lewis, 20 June 1785, draft, Blagden Letterbook, Yale. William Lewis to Charles Blagden, 25 June 1785, Blagden Letters, Royal Society, L.46.
The journal is in a wrapper labeled in Cavendish’s hand, “Computations & Observations in Journey 1785,” Cavendish Mss X(a) 4:8. The journal was written by Blagden, but the copy at Chatsworth is in a copyist’s hand. The original is in Blagden’s papers at Yale.
Middleton (1964, 172, 179, 189).
Gavin de Beer (1956, 3–4). George Shuckburgh (1777, 1–2, 12–13). William Roy (1777, 673). Henry Cavendish, “Rule for Taking Heights of Barometers,” Cavendish Mss VIII, 12; “Observations of Thermom. on Mont Blanc,” Cavendish Mss, Misc. Charles Blagden to Joseph Banks, 5 Oct. 1786, BL Add Mss 33272, pp. 19–20.
Middleton (1964, 132–133, 161).
Ibid., 9–11.
Blagden, Journal of 1785, p. 53.
Ibid., p. 57.
Charles Blagden to Joseph Banks, 31 July 1785, Banks Correspondence, Royal Botanic Gardens, Kew 1.199.
Initially there were problems with the piston rod and the sun-and-planet gear of Watt’s engine, but by early 1786 the repairs had been made. In 1789, a second engine was installed.
S.B. Hamilton (1958, 455–456).
Charles Blagden to Joseph Banks, 31 July 1785, Banks Correspondence, Royal Botanic Gardens, Kew, l.l99.
“Computations & Observations in Journey 1786,” Cavendish Mss X(a), 5. The wrapper is labeled in Cavendish’s hand; the narrative is written in the copyist’s.
Charles Blagden to C.J. Phipps, Lord Mulgrave, 2 Aug., 1786, draft, Blagden Letters, Royal Society 7:17. Charles Blagden to John Blagden Hale, 14 Sep. 1786, draft, ibid. 7:33. Charles Blagden to John Michell, 5 Aug. 1786, draft; in McCormmach (2012, 407–408).
2 Sep. 1786, Charles Blagden Diary, Yale, Osborn Shelves f c 16
Henry Cavendish, “Strata Which Michell Dug Through for Coal,” in Cavendish’s journal of the 1786 trip, Cavendish Mss X(a), 3:13–14.
Charles Blagden to C.J. Phipps, Lord Mulgrave, 2 Aug. 1786, draft, Royal Society 7:17.
“Examination of Substances Sent from Lord Mulgrave’s,” in “White Book,” Cavendish Mss Misc., pp. 7–13.
Kirwan thought that the phlogistication of air generates fixed air. Cavendish knew that it does not. Henry Cavendish (1784b, 184). In 1779 Scheele performed experiments on plumbago, a substance which had been used in pencils, showing that it consists mainly of carbon with some iron. Thomson (1830–1831, 2:71).
Peter Woulfe to Charles Blagden, 26 June [?] 1785, Blagden Letters, Royal Society W30.
Charles Blagden to John Michell, 19 Sep. 1786, draft; in McCormmach (2012, 409–412).
Charles Blagden to Joseph Banks, 17 Sep. 1786, BL Add Mss 33272, pp. 9–10.
Charles Blagden to Joseph Banks, 4 Sep. 1786, ibid., pp. 7–8.
Charles Blagden to Henry Temple, Lord Palmerston, 25 Nov. 1800, Blagden Papers, Yale, box 63/43.
Charles Blagden to Joseph Banks, 8[?] Oct. 1786, BL Add Mss, 33272, pp. 15–16.
Charles Blagden to James Watt, 23 Aug. 1787, draft, Blagden Letters, Royal Society 7:349. Two letters of introduction from George Hunt, 23 Jan. 1787, who was asked to write them by his nephew R. Wilbraham, “The bearers of this are Mr.Cavendish ….” Blagden Papers, Yale, box 1, folder 4. Along the way Blagden solicited letters: James Reynolds to Rev. Burlington, 18 Aug. 1787, “The bearer, Dr Blagden, is my particular friend ….” Blagden Letters, Royal Society, R.5.
Charles Blagden to Mrs. Grey, 14 June 1787, draft, Blagden Letters, Royal Society 7:324. Charles Blagden to William Watson, 22 Aug. 1787, draft, ibid. 7:347.
Josiah Wedgwood to Charles Blagden, 30 Dec. 1786, Gloucestershire Record Office, D 1086, F 158.
Thirty-page journal of the 1787 journey, by Blagden, in a copyist’s hand, and with many insertions in Cavendish’s hand. Cavendish Mss X(a), 6.
Charles Blagden to Mrs. Grey, 28 Aug. 1787, draft, Blagden Letters, Royal Society 7:351.
Charles Blagden to John Michell, 1 Sep. 1787, draft; in McCormmach (2012, 434–436).
Charles Blagden to William Lewis, 11 July 1787, draft, Blagden Letters, Royal Society 7:338.
Brian Le Messurier, ed. (1967, 15). Charles Blagden to William Farr, 12 June 1787, draft, Blagden Letters, Royal Society 7:67; and other correspondence with Farr around this time.
Henry Cavendish’s journal of the 1787 trip, Cavendish Mss X(a), 7.
There are several large sheets of observations taken with the barometer on the 1787 trip, in Cavendish Mss Misc.
Charles Blagden to Joseph Banks, 14 Aug. 1787, Add Mss 33272.
Charles Blagden to Joseph Banks, 13 July 1788, ibid.
Charles Blagden to Jean André Deluc, 5 Sep. 1789, draft, Blagden Letters, Royal Society 7:301.
Charles Blagden to Joseph Banks, 11 May 1793, BL Add Mss 33272, pp. 119–20. Henry Cavendish to Joseph Banks, 23 Sep. 1793; in Jungnickel and McCormmach (1999, 696).
Joseph Banks to Matthew Boulton, 6 and 18 July, 10 Aug. 1793, Birmingham Assay Office.
This twenty-one page paper on strata in Cavendish’s hand does not have a group number, but it is kept with the travel journals in the Cavendish Mss.
This book has 138 numbered pages; 90 loose sheets are laid between the bound ones. Large blank spaces are left in the book for cross referencing and later additions. It is a copy book for preserving results of experiments. “White Book No. 1,” Cavendish Mss, Misc. On p. 59 Cavendish refers to “2d book,” which suggests that there was once a “White Book No. 2.”
Henry Cavendish, “Paper Given to Cockshutt,” inserted loosely in “White Book No. 1,” Cavendish Mss Misc.
The guiding thought appeared in John Michell’s paper on earthquakes, where he noted that level countries show great expanses of the same strata: “we have an instance of this in the chalky and flinty counties of England and France, which (excepting the interruption of the Channel, and the clays, sands, of a few counties) compose a tract of about 300 miles each way.” John Michell (1760, 587).
Archibald Geikie, “Note on Cavendish as a Geologist,” in Cavendish, Sci. Pap. 2:432.
20 Feb. 1806, Certificates, Royal Society, 6.
Jean André Deluc (1809, vi, 24, 63–64). Deluc argued against Hall’s experimental conclusions, pp. 359–361. Kirwan said that geological facts are historical, relying on testimony, and that recourse cannot be made to experiment. Richard Kirwan (1799, 4–6, 482).
Playfair quoted in Deluc (1809, 11–14).
Henry Cavendish to John Michell, 27 May 1783, draft; in Jungnickel and McCormmach (1999, 567–569).
“Michell, John,” DNB, 1st ed. 13:333–334, on 334.
Wollaston’s father, Francis, born the same year as Cavendish and a classmate of Cavendish’s at Cambridge, took his degree in law but entered the Church instead. Skilled in astronomy, he had his own observatory and first-class instruments. With at least that much in common, on 8 Dec. 1768 Cavendish brought Francis Wollaston as a guest to a meeting of the Royal Society. The certificate proposing Wollaston’s membership is signed by Cavendish along with Maskelyne and several other prominent members. 3 Jan. 1769, Certificates, Royal Society 3:65. “Wollaston, Francis,” DNB, 1st ed. 21:778–779. One of Francis Wollaston’s sons, William Hyde Wollaston, was an eminent chemist, whom Cavendish proposed as he had his father for membership in the Royal Society. 9 May 1793, Certificates, Royal Society 5; “Wollaston, William Hyde,” DNB, 1st ed. 21:782–787, on 782. Another of Francis’s sons George Hyde Wollaston was one of Cavendish’s neighbors on Clapham Common, where Cavendish performed his experiments on the density of the Earth. George Hyde Wollaston’s house along with Cavendish’s are on the map of Clapham Common (Fig. 11.12). Another of Francis’s sons was Francis John Hyde Wollaston, Jacksonian Professor of Chemistry, from whom Cavendish received Michell’s apparatus. “Wollaston, Francis John Hyde,” DNB, 1st ed. 21:779–780. Michell’s association with the Wollastons went back as far as Cavendish’s. As a recently elected fellow of the Royal Society, Michell’s first recommendation for a new member, in 1762, was for Francis’s youngest brother, George Wollaston, then fellow and mathematical lecturer in Sidney-Sussex College, Cambridge. “Wollaston, Francis,” 779.
Henry Cavendish, “Paper Given to Maskelyne Relating to Attraction & Form of Earth,” VI(b), 1:19.
In more detail, his reasoning is as follows. He deduces the density of the Earth in two steps. The first step assumes the laws of pendulum motion. The second step assumes the inverse square law of gravitation. Step 1. Cavendish draws on two relations: the period of vibration of a pendulum is proportional to the square root of the length of the pendulum and inversely proportional to the square root of the restoring force on the pendulum. With the aid of an analogy between the horizontal torsion pendulum and an imagined vertical simple pendulum beating seconds, the length of which is known, Cavendish expresses the force required to move the small balls at the ends of the torsion arm, with its observed period of vibration, through any observed angle of deflection of the arm in terms of the weight of a ball. Step 2. Cavendish invokes Newton’s law of gravitation twice, once to express the attraction between a small ball and the nearby larger ball, or “weight,” and once to express the attraction between the small ball and the Earth. The latter attraction is written so as to include the to-be-determined average density of the Earth. Forming a ratio of the two attractions, he expresses the attraction of the “weight” on the ball in terms of the attraction of the Earth on the same ball. Finally, he combines Steps 1 and 2. The force of the twisted wire from Step 1 is equal to the force of attraction between the small balls and the “weights” from Step 2. By dividing one force by the other, Cavendish arrives at the desired result: the density of the Earth, expressed in terms of the density of water, is equal to a numerical factor times the square of the period of vibration of the torsion arm divided by the deflection of the arm. By means of this reasoning, Cavendish brings the world into his laboratory.
Isaac Newton (1962, 2:569–570). Cavendish stated the proportion as one part in fifty million, which applied to the 8-inch weights Michell intended to use. For the 12-inch weights Cavendish used, the proportion is roughly 3 times larger, but the order of magnitude of the minuteness remains the same.
Cavendish (1798, 250, 252, 254–255, 259, 263–267). C.W.F. Everett (1977, 548).
Cavendish (1798, 277, 280, 283–284).
Ibid., 284.
Charles Hutton to Henry Cavendish, 17 Nov. 1798; in Jungnickel and McCormmach (1999, 710–711).
Cavendish’s manuscript in the Royal Society does not show an interpolation on the last page. Perhaps Cavendish rewrote the last page, or perhaps he made no change in his wording in response to Hutton’s letter. Henry Cavendish to Charles Hutton, draft, n.d. [after 17 Nov. 1798]; ibid., 712.
Charles Hutton (1814, 2:64).
Kenelm Digby, 1645, quoted in “Biographical Memoir,” in William Gilbert (1958, xviii).
John Henry Poynting (1892, 565–566).
Stephen Toulmin and June Goodfield (1965, 125, 266).
Item 20 in A Catalogue of Sundry Very Curious and Valuable Mathematical, Philosophical, and Optical Instruments […] Of a Gentleman Deceased.… On Saturday the Fifteenth of June 1816, at Twelve O’clock,(London, 1816), Devon. Coll.
Playfair, quoted in Deluc (1809, 14–16).
Thomas Young (1807, 2:575).
Charles Blagden to Joseph Banks, 1 Apr. 1802, BL Add Mss 33272, pp. 172–173.
Boys is quoted by Clotfelter (1987, 211). Boys recommended using a room with a more uniform temperature then Oxford’s; his accuracy was great, despite his room.
Cavendish did not write an equation for the force of universal gravitation, as we do:
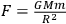 . He could have calculated G without having a unit of force, but he had no need for it, and it would not have occurred to him. Clotfelter (1987, 213).
. He could have calculated G without having a unit of force, but he had no need for it, and it would not have occurred to him. Clotfelter (1987, 213).
A.H. Cook (1987, 52). Appropriately, Cook talks of the Cavendish experiment only in connection with G and not with the density of the Earth. Only recently, he says, has the accuracy of G been improved upon over what can be obtained from Cavendish’s own experiment, and although in the study of materials we can achieve an accuracy of one part in 1012, we still know G only to about 1 part in 103.
Christian von Baeyer (1996, 98–99).
